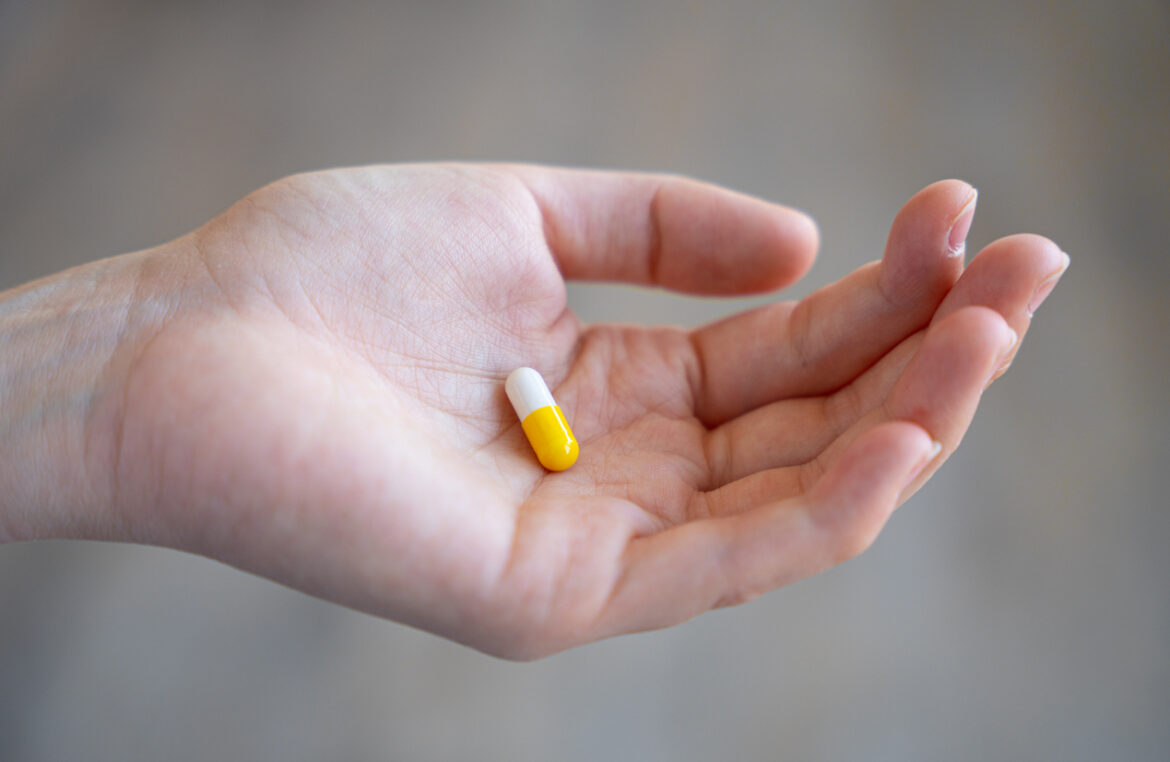
Amid rising cases of Covid-19 driven by the infectious Delta variant, oral coronavirus vaccine is set to start clinical trials in Israel, and the developer believes it could help get vaccines to countries that are struggling to inoculate because of limited infrastructure.
According to Eturbonews, Jerusalem-based Oramed Pharmaceuticals announced that it has received approval from the Tel Aviv Sourasky Medical Center to start a clinical trial on 24 unvaccinated volunteers for a single-dose capsule version of a COVID-19 vaccine.
READ: News: Study shows high exposure to malaria may reduce severe impact of COVID-19 on individuals
Oramed announced in March that it had tested its pill on pigs and the animals had produced antibodies after being administered.
The company which specializes in creating oral versions of drugs that are usually administered via an injection, is also currently conducting trials for its oral insulin capsule to treat Type-2 diabetes.
READ: Tourism: Six elderly tourists from South Africa die after 37 out of 40 on Namibian tour bus test positive for Covid
All currently existing COVID-19 vaccines are administrated via one or two injections.
According to Oramed CEO Nadav Kidron, the trial for the COVID-19 vaccine pill is expected to start next month, once it gains the final approval from the Health Ministry.
“Our oral vaccine, which doesn’t rely on a deep-freeze supply chain unlike other coronavirus vaccines, could mean all the difference between a country being able to emerge from the pandemic or not,” Kidron said.